Fabrication of magnetic amino-functionalized nanoparticles for S-arylation of heterocyclic thiols†
Abstract
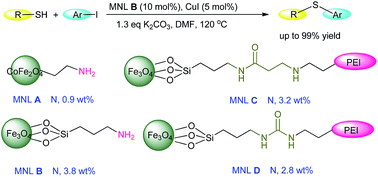
A series of uniformed mono-disperse magnetic nanoligands (MNLs) (CoFe2O4–NH2 (MNL A), Fe3O4@Si(CH2)3NH2 (MNL B), Fe3O4@Si(CH2)3NHC(O) (CH2)2PEI (MNL C) and Fe3O4@Si(CH2)3NHC(O)PEI (MNL D)) were obtained by loading two ligands, an aminosilane coupling agent and PEI-600, onto magnetic nanoparticles prepared using a solvothermal method. The catalytic applications of the synthesized MNLs were explored for the cross-coupling reaction of heterocyclic thiols with aromatic iodides. The reactions were carried out in the presence of CuI (5 mol%), MNL (10 mol% N) and K2CO3 (1.3 eq.) in DMF at 120 °C. A variety of heterocyclic sulfides were afforded in good to excellent yields (up to 98%) when MNL B was used. The magnetic, crystal, organic matter structure and morphology of MNL B exhibited no obvious changes after five consecutive cycles. XPS characterization of MNL B revealed the combination of a small amount of Cu0 nanoparticles, but this had no significant effect on catalytic performance.

 Please wait while we load your content...
Please wait while we load your content...