Electrochemical codeposition of graphene platelets and nickel for improved corrosion resistant properties
Abstract
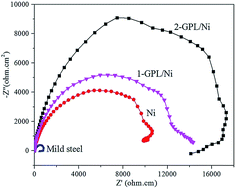
A study on graphene platelet (GPL)/nickel (Ni) composites was carried out to evaluate the potential of the new coating materials for corrosion resistance applications. Electrochemical deposition of Ni and GPL/Ni coatings on mild steel was performed in electrolytes with concentrations of 0, 0.1 and 0.2 g L−1 GPLs. Microstructures of the deposited coatings were examined using X-ray diffraction (XRD) and scanning electron microscopy. A Raman microscope was used to characterize the GPLs in the GPL/Ni composite coatings. Average grain sizes of composite coatings estimated from XRD were compared with that of pure Ni coating. Corrosion resistant properties of the coatings were examined by electrochemical corrosion tests. It is found that GPLs are well dispersed in the Ni matrix and agglomeration of GPLs occurs during the deposition process. The introduction of GPLs results in a significant refinement of the microstructure of the Ni matrix and a decrease of the texture coefficient of the Ni matrix at the preferred orientation. GPL/Ni composite coatings exhibit better corrosion resistance and higher microhardness than the pure Ni coating. In addition, considerable improvements in corrosion resistance and microhardness can be achieved by introducing a relatively higher content of GPLs. The research results prove a novel approach to construct corrosion resistant coatings using GPLs.

 Please wait while we load your content...
Please wait while we load your content...