Improvements in electrical and electrochemical properties of Nb-doped SnO2−δ supports for fuel cell cathodes due to aggregation and Pt loading†
Abstract
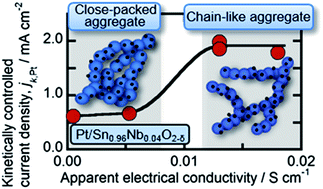
We found that Nb-doped SnO2−δ nanoparticles were improved in their electrical conductivity by more than two orders of magnitude by the aggregation of the particles, including the fusion of nearest-neighbor particles to form a random branching structure, and the formation of a porous agglomerated structure similar to that of conventional carbon blacks. The electrical conductivity was increased by an additional two orders of magnitude as a result of the loading of Pt nanoparticles to produce an electrocatalyst (Pt/Nb-doped SnO2−δ) for fuel cell applications. We also found that the specific activities of Pt/Sn0.96Nb0.04O2−δ (16 wt% Pt loading) for the oxygen reduction reaction increased with increasing electrical conductivity of the support and exceeded that of a conventional Pt catalyst supported on carbon black (Pt/CB). Since the tin oxide supports are much more stable than CB supports to the exposure to positive potentials (0.9 ∼ 1.3 V) during the start/stop operation of fuel cells, Pt or Pt alloy catalysts supported on oxide supports with such a chain-like, necked structure, as developed in this work, become superior candidates as robust cathode catalysts for the fuel cell vehicle application.

 Please wait while we load your content...
Please wait while we load your content...