The efficient hydroxyalkylation of phenol with formaldehyde to bisphenol F over a thermoregulated phase-separable reaction system containing a water-soluble Brønsted acidic ionic liquid†
Abstract
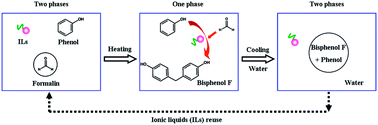
The efficient hydroxyalkylation of phenol with formaldehyde to bisphenol F over a thermoregulated phase-separable reaction system containing a water-soluble Brønsted acidic ionic liquid was studied. The reaction system containing the water-soluble IL showed thermoregulated biphasic behavior with change of the alkyl chain length of IL, temperature and water amount. Four types of imidazolium-, ammonium-, phosphonium- and pyridinium-based water-soluble ionic liquids with different anions of dihydrogen phosphate [H2PO4]−, acetate [CH3COO]− and hydrogen sulfate [HSO4]− were used as both Brønsted acidic catalysts and thermoregulated solvents. Among them, [C6MIM][HSO4] gave a high yield of 80.5% and a selectivity of 96.9% for bisphenol F, and the optimal reaction conditions were stirring speed 450 rpm, phenol–formaldehyde ratio 6 : 1, IL catalyst molar concentration 12.5%, reaction temperature 90 °C and reaction time 1 h. [C6MIM][HSO4] could be recovered by simple decantation and could retain its original activity even after six recycling-uses [CnMIM][HSO4] with the alkyl chain length n = 6 found to be the most suitable for the synthesis of bisphenol F because of both the formation of a thermoregulated monophasic reaction system at 90 °C to enhance the reaction efficiency and as a thermoregulated phase-transition solvent to facilitate its recovery from the reaction system.

 Please wait while we load your content...
Please wait while we load your content...