Programming galvanostatic rates for fast-charging lithium ion batteries: a graphite case
Abstract
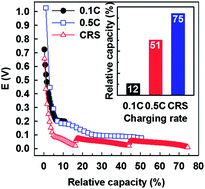
Galvanostatically induced lithiation of graphite, as a cathodic process of lithium ion batteries during charging, was investigated in situ by galvanostatic electrochemical impedance spectroscopy (GS-EIS). When lithiation is driven by charge rates slow enough for kinetics of the lithiation process to be considered relatively sluggish, charge transfer resistance (RCT) is slightly reduced as lithium ion intercalation proceeds from the dilute stage to stage 2L. Subsequently, RCT begins to increase during transformation of stage 2L to stage 2, followed by an abrupt increase in RCT observed during transition from stage 2 to stage 1, or after the inter-space of graphites is fully filled with lithium ions. As the ratio of charge rate to lithiated graphite increases, the potential responsible for the transition from stage 2L to stage 2 is shifted to more negative values due to significant polarization. Simultaneously, cells reach cut-off potentials before the transition from stage 2 to stage 1 proceeds. Based on the information regarding RCT profiles obtained by galvanostatic charging processes, a charging strategy is programmed with several different charge rates (C-rates). The capacity of lithiation is significantly enhanced by a C-rate switching (CRS) strategy. As a representative example, 75% of available capacity is charged for 50 minutes by a combination of 2 C, 1 C, and 0.5 C. However, only 12% and 51% of graphite is lithiated within the same time duration by a single charge rate of 0.1 C and 0.5 C, respectively.

 Please wait while we load your content...
Please wait while we load your content...