Facile microwave-assisted synthesis and effective photocatalytic hydrogen generation of Zn2GeO4 with different morphology†
Abstract
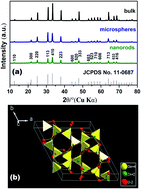
Single-crystalline hexagonal prism Zn2GeO4 nanorods and hierarchical Zn2GeO4 microspheres have been successfully synthesized via a facile microwave-assisted solution-phase approach. The as-prepared samples were characterized by XRD, SEM, TEM, HRTEM and UV-vis diffuse reflectance spectroscopy. The hierarchical Zn2GeO4 microspheres were found to be constructed of randomly aggregated nanorods which have dimensions of about 50 nm in length and 20 nm in width. Such rhombohedral phase Zn2GeO4 nanorods were found to be 10–20 nm in diameter and ∼200 nm in length. Some influencing factors such as the reaction time, temperature, and the urea were revealed to play crucial roles in the formation of Zn2GeO4 photocatalysts. A possible growth mechanism was proposed based on the experimental results. The rhombohedral phase Zn2GeO4 nanorods exhibited superior photocatalytic activities for the photocatalytic decomposition of water–methanol solution to hydrogen under UV irradiation.

 Please wait while we load your content...
Please wait while we load your content...