Electrochemical growth and characterization of a p-Cu2O thin film on n-ZnO nanorods for solar cell application
Abstract
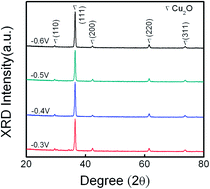
Heterostructural p-Cu2O/n-ZnO solar cells are fabricated by the consecutive electrochemical deposition of an n-ZnO nanorod layer, followed by a p-Cu2O layer on a transparent conductive substrate. The growth parameters, growth temperature and time of applied potential, which determine the crystalline structure and film thickness of Cu2O, are fine-tuned to investigate their effects on the performance of heterojunction solar cells. From XRD and SEM analyses, the best crystalline quality and the optimum Cu2O film thickness are accomplished at the potential of −0.4 V and the growth temperature of 60 °C for 10 min. In addition, the electrolyte concentration plays an important role in the optical absorbance of Cu2O film, which significantly exhibits two-time enhancement as adjusting the CuSO4 concentration of 0.4 M to 0.02 M. The best performance of the heterojunction cell is able to achieve the conversion efficiency of 0.33% with Voc, Jsc, and FF being 0.15 V, 7.03 mA cm−2, and 0.33, respectively. Hence, this study demonstrates that the proposed strategy to improve the solar cell performance realized by electrochemical deposition has the potential to produce cheap and environmentally friendly solar cells.

 Please wait while we load your content...
Please wait while we load your content...