Reversible Friedel–Crafts acyl rearrangements of planar polycyclic aromatic ketones: dibenzofluorenones†
Abstract
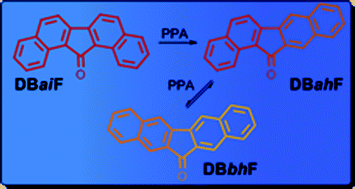
Dibenzofluorenones undergo reversible Friedel–Crafts acyl rearrangements in PPA at elevated temperatures. The Friedel–Crafts acyl rearrangements of 13H-dibenzo[a,i]fluoren-13-one (DBaiF) yield both 13H-dibenzo[a,h]fluoren-13-one (DBahF) and 12H-dibenzo[b,h]fluoren-12-one (DBbhF). DBahF and DBbhF undergo reversible mutual isomerizations, and their ratio depends on the reaction conditions. The O-protonate DBahFH+ plays a pivotal role in the proposed mechanism of the reversible Friedel–Crafts acyl rearrangements. DBahFH+ may undergo proton migration to give two isomeric σ-complexes: σ-13aH-DBahF+ and σ-12aH-DBahF+, leading, via the respective naphthyl naphthoylium ions βCOβN-βN+ and αCOβN-βN+ to O-protonates DBbhFH+ and DBaiFH+, respectively. The regioselectivity of the rearrangement is expressed by the preferred intramolecular beta-electrophilic attack in βCOβN-βN+ and by the preferred alpha-electrophilic attack in αCOβN-βN+, which indicates a thermodynamic control. The proposed mechanism is supported by the results of the DFT calculations of the dibenzofluorenones, their O-protonates, their σ-complexes and their corresponding naphthyl naphthoylium ions at B3LYP/6-311++G(d,p). DBahF and DBaiF are the kinetically controlled products of the Friedel–Crafts acyl rearrangement, while DBbhF is the thermodynamically controlled product. The aromaticity/antiaromaticity notions in dibenzofluorenones and their O-protonates, estimated by calculated HOMA and NICS indices, are discussed.

 Please wait while we load your content...
Please wait while we load your content...