Copper-catalyzed sequential arylation and intramolecular annulation of 2-(2-bromophenyl)-2,3-dihydroquinazolin-4(1H)-ones with amidines†
Abstract
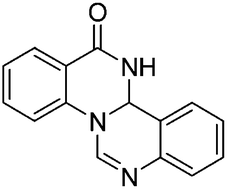
A copper-catalyzed sequential arylation and intramolecular annulation of 2-(2-bromophenyl)-2,3-dihydroquinazolin-4(1H)-ones with alkyl amidines has been developed for the synthesis of 6-alkyl 11bH-quinazolino[3,4-a]quinazolin-13(12H)-ones (3) in moderate to good yields. However, the arylated products N-(2-(4-oxo-1,2,3,4-tetrahydroquinazolin-2-yl)phenyl)benzimidamides (4) are isolated when aryl amidines are used in the transformation.

 Please wait while we load your content...
Please wait while we load your content...