Non-stoichiometry in “CaCu3Ti4O12” (CCTO) ceramics†
Abstract
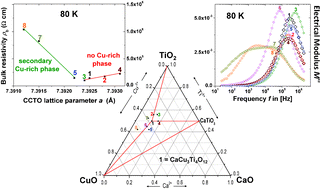
A combined powder X-ray lattice parameter and ceramic
Maintenance work is planned from 09:00 BST to 12:00 BST on Saturday 28th September 2024.
During this time the performance of our website may be affected - searches may run slowly, some pages may be temporarily unavailable, and you may be unable to access content. If this happens, please try refreshing your web browser or try waiting two to three minutes before trying again.
We apologise for any inconvenience this might cause and thank you for your patience.
* Corresponding authors
a
Universidad Complutense de Madrid, Departamento Física Aplicada III, GFMC, Facultad de Ciencias Físicas, 28040 Madrid, Spain
E-mail:
rainerxschmidt@googlemail.com
b The University of Sheffield, Materials Science and Engineering, Mappin Street, Sheffield, United Kingdom
c Indian Institute of Technology Kanpur, Department of Materials Science and Engineering, Kalyanpur, Kanpur - 208 016, India
d CNR-IMM, Strada VIII n.5, Zona Industriale, 95121 Catania, Italy
A combined powder X-ray lattice parameter and ceramic

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
R. Schmidt, S. Pandey, P. Fiorenza and D. C. Sinclair, RSC Adv., 2013, 3, 14580 DOI: 10.1039/C3RA41319E
This article is licensed under a Creative Commons Attribution 3.0 Unported Licence. You can use material from this article in other publications without requesting further permissions from the RSC, provided that the correct acknowledgement is given.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
