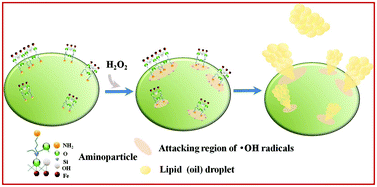
In this study, we demonstrated the synthesis of novel aminoparticles with Fe3+, Mn2+, and Cu2+ placed in active metal centers by means of a sol–gel reaction with 3-aminopropyltriethoxysilane (APTES) as a precursor, thereby forming –O(CH2)3NH2 organo-functionalities on cationic metals. The protonated amine groups led to the efficient flocculation of microalgae which occurred within as little as 5 min. To our surprise, the aminoparticles exhibited a Fenton-like activity when 1% hydrogen peroxide (H2O2) was introduced. Just like any Fenton-like reaction, it is plausible that •OH radicals are produced through the activation of H2O2 by the metal atoms embedded in the aminoparticles, bringing about cell damage and effectively releasing internal lipids from wet microalgae biomass. The efficiency of lipid extraction with the Fe-aminoparticle in the presence of 1% H2O2 was 26.70% and nearly 100% of the extracted oil was converted to biodiesel. It is believed that our multiple-functionality aminoparticles represent a promising means of substantially reducing the unit cost of microalgal biorefineries in general and microalgae-based biodiesel production in particular.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...