Improving sulfur tolerance of noble metal catalysts by tungsten oxide-induced effects†
Abstract
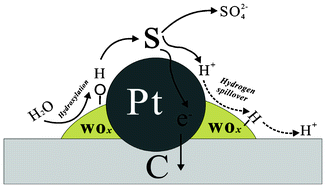
Sulfur adsorption and poisoning of Pt-based catalysts cause an undesired, detrimental effect on performance of proton exchange membrane fuel cells. Here, WOx is adopted for the first time to modify such noble metal catalysts with an aim of acquiring excellent sulfur tolerance, due to its unique nature with hydrophilicity, redox couple in lower valence, as well as proton spillover effect. A series of WOx–Pt/C catalysts with various contents of tungsten oxide from 1 to 50 wt% were synthesized and compared to conventional Pt/C catalysts toward sulfur resistance using cyclic voltammetry (CV), as well as rotating ring-disk electrode (RRDE) methods. The results show that the catalyst with 5 wt% WOx has excellent sulfur tolerance, which might be ascribed to the synergistic effect between WOx and Pt. The OH groups generated on hydrophilic surfaces of WOx would accelerate the oxidation of sulfur and lead to rapidly recovering the performance of poisoned WOx–Pt/C electrodes. The loss of SOx at the first potential cycling for poisoned WOx–Pt/C is as high as 51.2% of the total SOx coverage. Moreover, the higher catalytic activity of WOx–Pt/C toward oxygen reduction reaction (ORR) is revealed in comparison with Pt/C after both were poisoned by SOx where the electron transfer number of the former is closer to four-electron than that of the latter. The electronic interaction between Pt and WOx is evidently confirmed by X-ray photoelectron spectroscopy analysis and strongly suggested as the crucial factor for the ORR enhancement.

 Please wait while we load your content...
Please wait while we load your content...