Rh(i)-Catalyzed stereoselective intramolecular cycloaddition reactions of ene-vinylidenecyclopropanes for the construction of fused 6,5-bicyclic skeletons with a quaternary all-carbon stereocenter†
Abstract
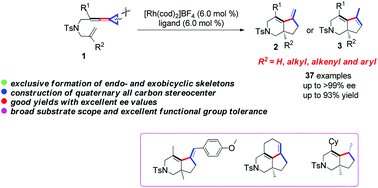
A cationic Rh(I) complex catalyzed intramolecular cycloaddition reaction of ene-vinylidenecyclopropanes to produce bicyclo[4.3.0] skeletons containing a quaternary all-carbon center at the ring juncture has been successfully established. The reaction could afford a range of desired bicyclic products in good yields with a broad substrate scope and good functional-group tolerance. The asymmetric variant of this cycloaddition could also be achieved by using chiral bisphosphine ligands to give the desired products with high ee values. Furthermore, the exclusive production of endo- and exocyclic double bond products could be achieved when the substituent at the quaternary carbon center is a methyl group. The synthetic utility of the obtained products has been demonstrated by several transformations of the obtained bicyclic products. The reaction mechanism and product selectivity were studied by control experiments, and plausible reaction mechanisms have been proposed.


 Please wait while we load your content...
Please wait while we load your content...