Synthesis of trifluoromethylated 3,4-dihydroquinolin-2(1H)-ones via a photo-induced radical cyclization of benzene-tethered 1,7-enynes with Togni reagent†
Abstract
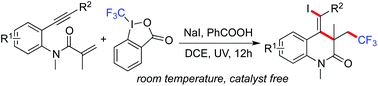
A photoinduced radical cyclization of benzene-tethered 1,7-enynes with Togni reagent in the presence of sodium iodide is developed. Under ultraviolet irradiation, (Z)-4-(iodomethylene)-3-(2,2,2-trifluoroethyl)-3,4-dihydroquinolin-2(1H)-ones are generated in moderate to good yields. The transformation works well without any metals or photo-redox catalysts at room temperature. During the reaction process, the reaction proceeds through a trifluoromethyl radical-triggered α,β-conjugated addition/intramolecular 6-exo cyclization/iodination, with the formation of multiple bonds. Good functional group tolerance is observed under the reaction conditions.

 Please wait while we load your content...
Please wait while we load your content...