Hexagonal tungsten oxides with large bandgaps synthesized by a chemical substitution method†
Abstract
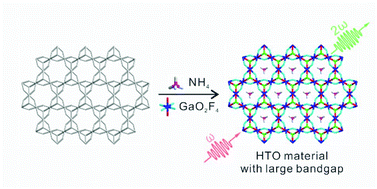
Two novel hexagonal tungsten oxide materials, A(GaF2)3(SeO3)2 (A = K and NH4), designed by a chemical substitution-oriented method have been successfully synthesized via hydrothermal reactions. A(GaF2)3(SeO3)2 feature two-dimensional corrugated layered structures composed of GaO2F4 octahedra, SeO3 trigonal pyramids, and A+ counter cations. Although the title compounds are stoichiometrically equivalent, K(GaF2)3(SeO3)2 crystallizes in the centrosymmetric trigonal space group, R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, whereas NH4(GaF2)3(SeO3)2 crystallizes in the noncentrosymmetric hexagonal space group, P63mc. UV-vis diffuse reflectance spectroscopic analysis indicates that the optical bandgaps of K(GaF2)3(SeO3)2 and NH4(GaF2)3(SeO3)2 are very large with the values of ca. 5.62 and 5.77, respectively. Noticeably, NH4(GaF2)3(SeO3)2 exhibits the largest bandgap among all known phase-matchable nonlinear optical selenite compounds. Powder second-harmonic generation (SHG) measurements indicate that NH4(GaF2)3(SeO3)2 reveals an SHG efficiency of ca. 1.1 times that of KDP and is type-I phase-matchable. Complete spectroscopic and thermal characterization, calculations, and factors influencing the macroscopic centricity will be discussed.
m, whereas NH4(GaF2)3(SeO3)2 crystallizes in the noncentrosymmetric hexagonal space group, P63mc. UV-vis diffuse reflectance spectroscopic analysis indicates that the optical bandgaps of K(GaF2)3(SeO3)2 and NH4(GaF2)3(SeO3)2 are very large with the values of ca. 5.62 and 5.77, respectively. Noticeably, NH4(GaF2)3(SeO3)2 exhibits the largest bandgap among all known phase-matchable nonlinear optical selenite compounds. Powder second-harmonic generation (SHG) measurements indicate that NH4(GaF2)3(SeO3)2 reveals an SHG efficiency of ca. 1.1 times that of KDP and is type-I phase-matchable. Complete spectroscopic and thermal characterization, calculations, and factors influencing the macroscopic centricity will be discussed.


 Please wait while we load your content...
Please wait while we load your content...