Thin-walled hollow Au–Cu nanostructures with high efficiency in electrochemical reduction of CO2 to CO†
Abstract
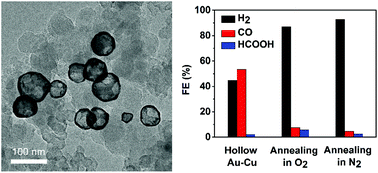
Bimetallic hollow structures can be efficiently used to improve the utilization of noble metals in electrocatalysts, but few studies have reported the application of bimetallic hollow nanocomposites for the electrochemical reduction of CO2. Herein, we demonstrated the synthesis of hollow Au–Cu nanoparticles of size ranging from 50 nm to 100 nm with a wall thickness of 4.95 ± 0.81 nm for efficient CO2 reduction reaction (CO2RR) via galvanic replacement and the Kirkendall effect between copper and gold. In the fabrication procedure, a gold precursor was pre-dissolved in TOP to lower the reduction potential of Au3+, which could then be reduced by Cu0. The obtained hollow Au–Cu NPs were then supported on carbon black for CO2RR measurement, and it exhibited a maximum CO FE of 53.3% at −0.7 V (vs. RHE), which was very close to that of Au NP/C, but the gold mass current density of hollow Au–Cu NP/C was much higher. The hollow Au–Cu NP/C also showed good stability in long-time measurements for CO2RR. To find out how the superior catalytic performance was achieved, hollow Au–Cu NP/C was then annealed in O2 and N2. It was found that after being annealed, the catalysts almost totally lost the activity towards CO2RR, which may be due to the partial segregation of Cu species on the nanoparticle surface as well as a decrease in the surface defect density with the improvement in crystallinity. Thus, it was concluded that high dispersion of more Au atoms on the nanoparticle surface together with the presence of more surface defect sites could account for the superior CO2RR performance of the as-synthesized hollow Au–Cu NP/C.
- This article is part of the themed collections: Celebrating 110th Anniversary of Chemistry at Peking University and Inorganic Chemistry Frontiers HOT articles for 2018


 Please wait while we load your content...
Please wait while we load your content...