A series of porous interpenetrating metal–organic frameworks based on fluorescent ligands for nitroaromatic explosive detection†
Abstract
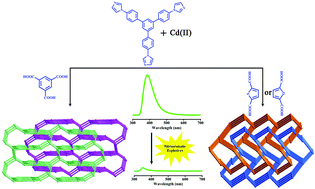
Four porous interpenetrating metal–organic frameworks (MOFs), namely, [Cd2(Tipb)2(btc)Cl]·Gx (1), [Cd(Tipb)(tdc)]·Gx (2), [Cd(Tipb)(fdc)]·Gx (3), and [Cd5(Tipb)4(btc)2Cl2(DMF)2(H2O)5]·Cl2·Gx (4) (G = guest molecules, Tipb = 1,3,5-tris(p-imidazolylphenyl)benzene, btc = benzene-1,3,5-tricarboxylic acid, tdc = thiophene-2,5-dicarboxylic acid, fdc = furan-2,5-dicarboxylic acid, and DMF = N,N′-dimethylformamide) have been assembled from π-electron-rich ligands and cadmium salts. These four MOFs exhibit strong fluorescent emissions upon excitation at room temperature. Due to the presence of π-electron-rich N-donor ligands, the sensing sensitivities of compounds 1–3 towards electron-deficient nitroaromatic explosives such as 2,4,6-trinitrophenol (TNP), 2,4-dinitrotoluene (2,4-DNT), 1,3-dinitrobenzene (1,3-DNB), and nitrobenzene (NB) have been studied by fluorescence quenching experiments. The results indicate that compounds 1–3 are highly sensitive to TNP owing to the electron and energy transfers between MOFs and TNP. Furthermore, compounds 1–3 remain stable after quenching and exhibit good recyclability. Moreover, the adsorption properties of 1–3 have also been investigated.


 Please wait while we load your content...
Please wait while we load your content...