Structural and magnetic susceptibility characterization of Pu(v) aqua ion using sonochemistry as a facile synthesis method†
Abstract
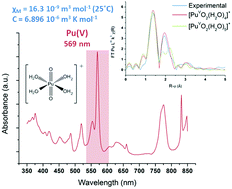
Since the past few years, Pu(V) has gained much attention due to its potential contribution to the environmental migration of actinides. However, the preparation of concentrated (up to mM) and pure Pu(V) solutions is quite difficult and often hindered by its great instability towards disproportionation, thus limiting the accessibility to physical and chemical property data. This work describes the rapid and facile sonochemical preparation of relatively stable Pu(V) solutions in the millimolar range free from the admixtures of the other oxidation states of plutonium. The mechanism deals with the sonochemical reduction of Pu(VI) in weakly acidic perchloric solutions by using the in situ generated H2O2, where the kinetics can be dramatically enhanced under high frequency ultrasound and an Ar/O2 atmosphere. The quasi-exclusive presence of the Pu(V) aqua ion in solution was evidenced by UV-vis absorption spectroscopy. The prepared solutions were found to be stable for more than one month which allowed the accurate XAFS and NMR investigations of Pu(V). EXAFS spectra revealed the presence of two trans dioxo Pu![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds at 1.81 Å and 4–6 equatorial Pu–Oeq interactions at 2.47 Å characteristic of coordinated water molecules. The exact number of water molecules (N[Oeq(H2O)] = 4) was determined by simulating the EXAFS spectra of the PuO2+ aqua complexes using DFT calculations (geometry and the Debye–Waller factor) and comparing them with experimental signals. For the first time, the magnetic susceptibility of the pentavalent state of plutonium in aqueous solutions was also determined (χM = 16.3 × 10−9 m3 mol−1 at 25 °C) and the related Curie constant was estimated (C = 6.896 × 10−6 m3 K mol−1).
O bonds at 1.81 Å and 4–6 equatorial Pu–Oeq interactions at 2.47 Å characteristic of coordinated water molecules. The exact number of water molecules (N[Oeq(H2O)] = 4) was determined by simulating the EXAFS spectra of the PuO2+ aqua complexes using DFT calculations (geometry and the Debye–Waller factor) and comparing them with experimental signals. For the first time, the magnetic susceptibility of the pentavalent state of plutonium in aqueous solutions was also determined (χM = 16.3 × 10−9 m3 mol−1 at 25 °C) and the related Curie constant was estimated (C = 6.896 × 10−6 m3 K mol−1).


 Please wait while we load your content...
Please wait while we load your content...