Structural stability and thermoelectric properties of cation- and anion-doped Mg2Si0.4Sn0.6†
Abstract
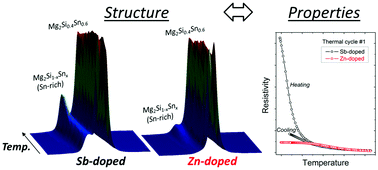
Undoped, anion-doped (Sb, Bi), and cation-doped (Ca, Zn) solid solutions of Mg2Si0.4Sn0.6 have been prepared by a commercially feasible large-scale solid state synthesis method. The compositional and structural stability of the prepared samples are investigated by high resolution synchrotron powder X-ray diffraction (PXRD) in the potential application temperature range of 300–750 K. Quantitative compositional and structural information are extracted from the multi-temperature PXRD data by the Rietveld method. Detailed analysis of the PXRD data reveals an irreversible thermally induced partial conversion of Mg2Si0.4Sn0.6 into a discrete Sn-rich Mg2Si1−xSnx-phase in the undoped and anion-doped samples. On the other hand, the cation-doped samples only undergo very minor compositional and structural changes with increasing temperature, indicating a stabilizing effect of Ca and Zn on the Mg2Si0.4Sn0.6 solid solution. The structural instability of the undoped and anion-doped samples is corroborated by the measured electrical resistivity as function of temperature in the same temperature range, in which a clear difference is observed between values during initial heating and subsequent cooling. In contrast, the resistivity data of the cation-doped samples exhibit good repeatability for two thermal cycles, confirming that cation doping greatly improves the thermal stability. This work highlights the importance of conducting multiple temperature cycles in the measurement of physical properties combined with a thorough structural characterization in studies of thermoelectric materials.
- This article is part of the themed collections: In honour of Mercouri G. Kanatzidis for his contributions to Inorganic Chemistry for over 30 years and Inorganic Chemistry Frontiers HOT articles for 2017


 Please wait while we load your content...
Please wait while we load your content...