Effect of the conjugation pathway on the electronic structures of p–π* conjugated polymers with fused borepin units†
Abstract
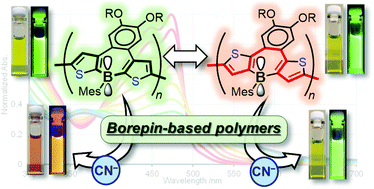
Among conjugated materials, p–π* conjugated polymers have attracted much attention due to their unique electronic structures derived from the orbital interaction between the empty p-orbital on boron and the π*-orbitals of the adjacent π-systems. On the other hand, borepin, an isoelectronic ring system of tropylium ions, has been investigated as an aromatic system containing heteroatoms. In this work, the first examples of p–π* conjugated polymers consisting of tetracyclic borepin structures with different conjugation pathways were prepared. Optical investigations revealed that the borepin polymers have more extended conjugations than conventional p–π* conjugated polymers. Furthermore, the two borepin polymers exhibited drastically different optical responses when cyanide was added to the solution. Photophysical measurements and DFT calculations disclosed the characteristic electronic effects of the borepin structures and the influence of different conjugation pathways.


 Please wait while we load your content...
Please wait while we load your content...