Self-assembly of supramolecular chemoenzymatic poly-l-phenylalanine†
Abstract
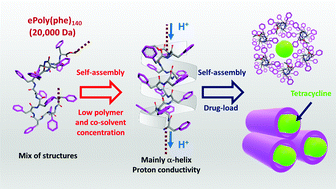
The self-assembly behavior of chemoenzymatic high molecular weight (ca. 30 000 Da) poly-L-phenylalanine (ePLP) and the nano-morphologies thereof are investigated. Interestingly, this supramolecular ePLP changes from a non-ordered secondary structure when solubilized in water to an α-helix with the addition of only 1 vol% HFIP co-solvent, and at a relatively low concentration (0.5 mg mL−1). The results also indicate a temperature-dependent structural stabilization, wherein the self-assembly process is mainly governed by hydrogen bonding. The analyses evidence self-assembly under controlled conditions to obtain nanotubes with an average diameter of 0.21 ± 0.04 μm, and above 100 μm length. Microbeam synchrotron radiation infrared (SRIR) spectroscopy demonstrates parallel orientations of the amide moieties in the α-helical structure, which is extraordinary for this long chain ePLP. The achievement of these highly homogeneous nanotubes is relevant since previous reports only demonstrated this arrangement for end-capped F-moc-containing short oligophenylalanines. In the same way, the conductivity behaviors of the nanotubular structure remained unchanged in the range of 273–313 K demonstrating its stability from hydrogen bonds, and also, direct dielectric measurements reveal a high orientation and stabilization by intra- and intermolecular hydrogen bonds in the nanotubes. These characteristics allow drug delivery assays with tetracycline-loaded nanotubes and further applications are envisaged.


 Please wait while we load your content...
Please wait while we load your content...