Aminolysis induced functionalization of (RAFT) polymer-dithioester with thiols and disulfides†
Abstract
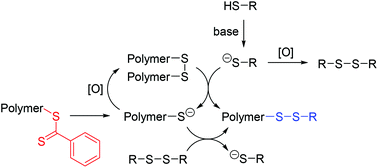
A series of polystyrene- and poly(methyl methacrylate)-dithioesters was subjected to aminolysis under ambient atmospheric conditions, i.e., in the presence of oxygen. Polymer disulfide coupling by oxidation occurred within tens of minutes and the yield of disulfide-coupled polymer increased with decreasing polymer molar mass. Oxidation of thiolates is usually an unwanted side reaction, here it is employed to obtain exclusively polymeric mixed disulfides through in situ aminolysis/functionalization in the presence of a thiol. The in situ aminolysis/functionalization in the presence of a disulfide, Ellman's reagent or polymer disulfide, resulted in the exclusive formation of polymer-dithionitrobenzoic acid, which can be further reacted with a thiol to exchange the terminal functionality, or block copolymer with dynamic disulfide linker, respectively.


 Please wait while we load your content...
Please wait while we load your content...