The difluoromethylthio moiety lowers the LCST of oligo(ethylene glycol)-based homopolymers†
Abstract
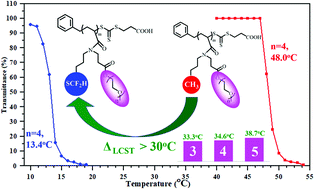
A series of thermo-responsive homopolymers bearing difluoromethylthio and oligo(ethylene glycol) moieties in each repeat unit were firstly synthesized from newly synthesized acrylamide monomers, N-3-(difluoromethylthio)propyl-N-(3-methoxyl-(ethoxy)n-3-oxopropyl)acrylamide, that is, DFTP-MEOn-AM (n = 2–5). These new DFTP-MEOn-AM monomers were prepared from a key amine intermediate bearing difluoromethylthio through aza-Michael addition reaction followed by amidation with acryloyl chloride. Well-defined poly(DFTP-MEOn-AM) homopolymers obtained via reversible addition–fragmentation chain transfer (RAFT) polymerization exhibited good temperature-responsive properties. The LCSTs of poly(DFTP-MEOn-AM) homopolymers are ∼35 °C lower than those of corresponding poly(Bu-MEOn-AM) homopolymers without fluorine, which demonstrated that introduction of difluoromethylthio moiety could effectively lower LCST of related polymers. LCSTs of poly(DFTP-MEO4-AM) homopolymers were also affected by isotopic solvent, polymer concentration, salt, urea, etc. Temperature-variable 1H NMR analysis indicated that difluoromethylthio moiety could affect the dehydration process of poly(DFTP-MEO4-AM) homopolymer above LCST.


 Please wait while we load your content...
Please wait while we load your content...