Aqueous solution behavior of stimulus-responsive poly(methacrylic acid)-poly(2-hydroxypropyl methacrylate) diblock copolymer nanoparticles†
Abstract
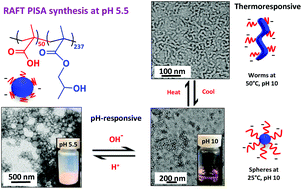
Poly(methacrylic acid)-poly(2-hydroxypropyl methacrylate) (PMAA50-PHPMA237) diblock copolymer nanoparticles are synthesized via reversible addition–fragmentation chain transfer (RAFT) aqueous dispersion polymerization, which is an example of polymerization-induced self-assembly (PISA). These nanoparticles exhibit complex stimulus-responsive behavior in dilute aqueous solution. They undergo macroscopic precipitation at low pH owing to protonation of the PMAA steric stabilizer. However, adjusting the solution pH above the pKa of 6.3 for the PMAA block ensures colloidal stability and confers thermoresponsive behavior. The degree of ionization of these anionic stabilizer chains increases at high pH, which leads to increasingly negative zeta potentials as judged by aqueous electrophoresis. Variable temperature dynamic light scattering (DLS) studies indicate the formation of progressively larger nanoparticles at higher temperatures, with TEM images providing evidence for weakly anisotropic nanoparticles at 50 °C. These observations are consistent with variable temperature 1H NMR spectroscopy studies, which indicate gradual dehydration of the structure-directing PHPMA block. Rheology measurements on a 20% w/w copolymer dispersion indicate a critical gelation temperature of around 10 °C and a gel modulus (G′) of approximately 1000 Pa at 25 °C. Shear-induced polarized light imaging (SIPLI) studies confirm the presence of weakly anisotropic worm-like particles under such conditions.


 Please wait while we load your content...
Please wait while we load your content...