Orthogonally functionalizable polyacetals: a versatile platform for the design of acid sensitive amphiphilic copolymers†
Abstract
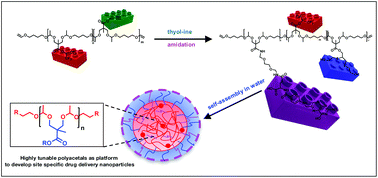
To expand the range of hydrolytically degradable functional polymers, copolyacetals of various compositions were synthesized and fully characterized, and their self-assembly into nanoparticles was studied. First, functionalized copolyacetals were obtained by polyaddition of alkyne- and activated ester-functionalized diols with 1,4-butanediol divinyl ether (DVE). Second, the copolymers were functionalized by using two orthogonal chemical reactions, the thiol–yne click reaction and transesterification/amidation. Amphiphilic copolymers of varying compositions, i.e. different hydrophilic/hydrophobic balance, were obtained and self-assembled in water to afford well-defined nanostructures. The size of the micelles and the critical micelle concentration were correlated to the chemical nature and to the modification degree of the copolymers. The micelles displayed high drug loading capacity and encapsulation efficiency and could be disassembled in mildly acidic media showing a pH-dependent drug release behavior. Subsequent orthogonal modification to introduce folic acid moieties allowed the preparation of site-specific drug delivery systems, showing the versatility of this approach.


 Please wait while we load your content...
Please wait while we load your content...