Engineering mannosylated nanogels with membrane-disrupting properties†
Abstract
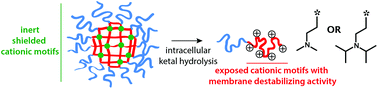
In this work, mannosylated core-cross-linked nanogels are designed that contain cationic moieties in their core. Mannosylated systems are attractive for targeting receptors on immune cell subsets that recognize structures with multiple mannose motifs. In the intact state, these nanogels are intert, but upon pH-triggered hydrolysis of a ketal motiety in the cross-links, the nanogels disassemble into soluble unimers and exert potent membrane-destabilizing activity. The latter is an important feature in view of cytoplasmic delivery of membrane impermeable (macro)molecules. At lower pH, N,N-dimethylamine and N,N-diisopropylamine bear a cationic charge, with N,N-diisopropylamine showing the highest membrane-destabilizing activity which is attributed to a combination of electrostatic charge interaction between the amines and the negative phospholipid membrane and hydrophobic interaction fostered by the diisopropyl moieties.


 Please wait while we load your content...
Please wait while we load your content...