Enhanced water transport in AEMs based on poly(styrene–ethylene–butylene–styrene) triblock copolymer for high fuel cell performance†
Abstract
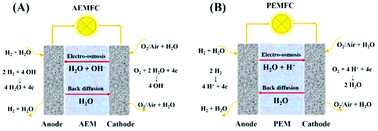
Anion exchange membrane fuel cells (AEMFCs) have received a considerable amount of attention in the past decades as a lower cost alternative to proton exchange membrane fuel cells (PEMFCs). However, limitations in performance and stability have hindered the AEMFC development, and poor water management was a major reason. In the AEMFCs, water is both electrochemically generated at the anode and consumed at the cathode during cell operation, so water transport is particularly important because of its severe intrinsic imbalance. In this work, a high conductivity and stability anion exchange membrane and a soluble, conductive ionomer based on triblock copolymer poly(styrene–ethylene–butylene–styrene) (SEBS) were synthesized successfully. The OH− conductivity of the AEM was over 0.1 S cm−1 at 80 °C. Remarkably, the AEM could be stable in 1 M KOH at 60 °C for over 1700 h, and the conductivity decreased only 26% (from 60.77 to 44.91 mS cm−1 at 30 °C). Moreover, the QASEBS AEM exhibited a good water transport property in ex situ characterization, which contributed to improving water management in AEMFCs. Besides, H2/O2 fuel cells with the AEMs and ionomer were operated at 60 °C, and the results showed that the cathode dew points and the thickness of the AEMs played a key role in the AEMFC performance, and the cathode unsaturated humidification was conducive to improving the performance, which was ascribed to the fact that the increase of the concentration gradient of water between the anode and the cathode contributed to the water back diffusion from the anode to the cathode. Moreover, the back diffusion not only relieved the flooding at the anode, but replenished water consumed at the cathode. After optimization, the AEMFC performance reached 721.7 mW cm−2 at the cell temperature of 60 °C and the anode/cathode dew point of 60 °C/50 °C.


 Please wait while we load your content...
Please wait while we load your content...