In vivo programming of tumor mitochondria-specific doxorubicin delivery by a cationic glycolipid polymer for enhanced antitumor activity†
Abstract
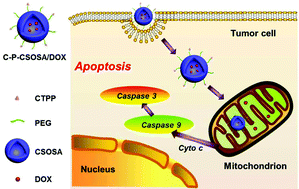
The selective delivery of DOX into tumor mitochondria in vitro and in vivo, which can be achieved with the aid of a skeletal polymer with positively charged and lipophilic character, may significantly promote its antitumor efficiency as well as eliminate its severe cardiotoxicity. In this study, we developed a polycationic glycolipid polymer (CSOSA) as a skeletal nanomaterial, which was further modified with lipophilic (4-carboxybutyl) triphenylphosphonium bromide (CTPP) cations to form the mitochondria-targeted polymer C-P-CSOSA. In vitro, C-P-CSOSA polymer micelles were internalized more by tumor cells than normal cells and exhibited rapid lysosomal escape capability due to their strong proton buffering ability. When loaded with DOX, the C-P-CSOSA/DOX micelles could selectively target mitochondria in tumor cells and remarkably activate the mitochondrial apoptosis signaling pathway. Importantly, the C-P-CSOSA polymer exhibited significant mitochondrial co-localization in vivo. Moreover, C-P-CSOSA/DOX demonstrated the highest tumor-inhibition rate (75.0%), when compared with CSOSA/DOX (44.8%) and DOX·HCl (55.0%). Meaningfully, C-P-CSOSA/DOX did not cause obvious organ damage and exhibited the longest survival time, indicating the low toxicity of this polymer. This study opens up new avenues for the future clinical translation of mitochondrial-targeted drug delivery systems.


 Please wait while we load your content...
Please wait while we load your content...