The synergistic effect during biphasic SET-LRP in ethanol–nonpolar solvent–water mixtures
Abstract
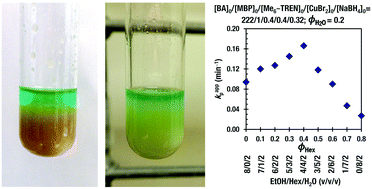
Single electron transfer-living radical polymerization (SET-LRP) of n-butyl acrylate (BA) catalyzed by Cu(0) generated via in situ reduction of Cu(II)Br2 with NaBH4 proceeds in triphase in ethanol–water mixtures since poly(butyl acrylate) (PBA) is not soluble in ethanol beyond a certain molecular weight and concentration. This triphase consists of the PBA gel swelled in the organic phase and the water phase containing Cu(II)Br2 and hexamethylated tris(2-aminoethyl)amine (Me6-TREN). Addition of at least 10% hexanes or other nonpolar and non-disproportionating solvents to the ethanol–water reaction mixture transforms SET-LRP of BA from a triphasic to a biphasic system. A synergistic effect was observed in the biphasic system, which was caused by the addition of hexanes that corresponds to a volumetric ratio of 4/3/3 for ethanol/hexanes/water, when the volume fraction of hexanes (ϕHex) is constant at 0.3, and 4/4/2 while the volume fraction of water (ϕH2O) is constant at 0.2. Other nonpolar, non-disproportionating solvents such as ethyl acetate, toluene, anisole, diethyl carbonate and cyclohexane were all shown to mediate the SET-LRP of BA in ethanol–nonpolar solvent–water mixtures at a 4/4/2 (v/v/v) ratio. These experiments demonstrate for the first time the ability to use a diversity of non-disproportionating, nonpolar solvents in conjunction with miscible polar solvents and water in biphasic SET-LRP. This new methodology is important from mechanistic and also from technologic points of view since it expands the scope of polymer synthesis via SET-LRP from disproportionating to non-disproportionating nonpolar solvents.

 Please wait while we load your content...
Please wait while we load your content...