Polyhydroxybutyrate (PHB)-based triblock copolymers: synthesis of hydrophobic PHB/poly(benzyl β-malolactonate) and amphiphilic PHB/poly(malic acid) analogues by ring-opening polymerization†
Abstract
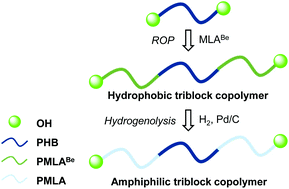
Polyhydroxyalkanoates (PHAs) are biocompatible and biodegradable polyesters widely used for biomedical applications. Amphiphilic triblock copolymers with a PHB hydrophobic segment recently demonstrated attractive advantages such as high colloidal stability and low critical micellar concentration (CMC) values for the elaboration of drug delivery systems. Our approach aims at associating PHB with a fully biodegradable hydrophilic block to access new self-assembled systems with unique potential. Well-defined α,ω-dihydroxy telechelic PHA-based hydrophobic triblock copolymers with tunable segments length were synthesized by the controlled ring-opening polymerization (ROP) of benzyl β-malolactonate, using polyhydroxybutyrate (PHB) diol/Nd(OTf)3 as the catalytic system. Remarkably, the reaction proceeds with the selective oxygen–acyl bond cleavage of the β-lactone. The corresponding amphiphilic copolymers were then obtained by hydrogenolysis. These copolymers are the only examples of fully biodegradable PHA-PHB-PHA triblock copolymers. The hydrophilic weight fraction of the copolymers was tuned from 7 to 83% upon modulating the monomer loading thus affording the ability to next access different self-assembling architectures.

 Please wait while we load your content...
Please wait while we load your content...