Polymerization and degradation of aliphatic polyesters synthesized by atom transfer radical polyaddition†
Abstract
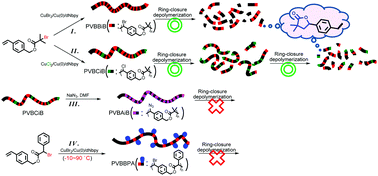
Two AB*-type inimers (4-vinylbenzyl 2-bromo-2-isobutyrate (VBBiB) and 4-vinylbenzyl 2-bromo-2-phenylacetate (VBBPA)) were synthesized and used to obtain aliphatic polyesters through atom transfer radical polyaddition (ATRPA). First, we optimized the ATRPA of VBBiB at a moderate temperature (<25 °C) using a commercially available CuBr2/Cu(0)/dNBpy system; however, the reaction run at 70 °C exhibited self-degrading behavior, as revealed by molecular weight (MW) variations in GPC traces and evidence of a five-membered-ring lactone structure (5-(4-(bromomethyl)phenyl)dihydro-3,3-dimethylfuran-2(3H)-one) in 1H NMR spectra. We suspected that the structural rearrangement occurred through ring-closure depolymerization of PVBBiB induced by the pendant bromine atoms serving as leaving groups. Accordingly, by varying the leaving group affinities, we performed a series of model reactions that guided us toward polymers with stable backbones. We thus synthesized three additional aliphatic polyesters PVBCiB (C: chloro), PVBBPA, and PVBAiB (A: azido) through ATRPA of VBBiB with halogen exchange, ATRPA of VBBPA, and azido substitution of bromine atoms, respectively. We confirmed the aliphatic polyester structures of PVBCiB and PVBBPA with high MWs (e.g., Mw,PVBBPA = 25 550) which fulfilled our desire to obtain high-MW aliphatic polyesters through ATRPA method. From a comparison of the four aliphatic polyesters, PVBBiB and PVBCiB displayed significant self-degrading profiles over periods of several days, whereas PVBBPA and PVBAiB did not. This study unveils the limitations (relating to lactonization) of ATRPA involving AB*-type inimers and also widens the scope of the process.

 Please wait while we load your content...
Please wait while we load your content...