Group 4 metal complexes bearing the aminoborane motif: origin of tandem ring-opening metathesis/vinyl-insertion polymerization†
Abstract
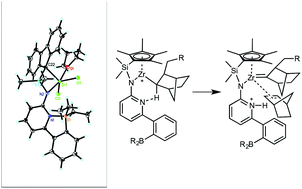
Three tailored ansa-type Zr(IV)- and Hf(IV)-complexes, Zr-1, Hf-1 and Zr-2, all bridged by a dimethylsilylene group and bearing both a 6-[2-(BR2)phenyl]pyrid-2-yl motif (R = ethyl, mesityl) and an η1/η5-bound ligand with different Lewis base character and steric demand, have been synthesized. Their structures have been determined by single-crystal X-ray diffraction analysis. Upon activation with methylalumoxane (MAO), Zr-1, Hf-1 and Zr-2 are capable of polymerizing norborn-2-ene (NBE) via ring-opening metathesis polymerization (ROMP). In the MAO-activated copolymerization of ethylene (E) with NBE; however, pure vinyl insertion polymerization (VIP)-derived poly(NBE)-co-poly(E) is obtained by Zr-1, Hf-1 and the model pre-catalyst (Zr-3), which does not contain the borylamino motif. In contrast, Zr-2 yields copolymers containing both ROMP- and VIP-derived poly(NBE)ROMP-co-poly(NBE)VIP-co-poly(E) units via an α-H elimination process. Variable-temperature 11B NMR measurements allow for identifying tri- and tetracoordinated borane species. The propensity of catalysts Zr-1, Hf-1, Zr-2 and Zr-3 to switch from VIP to ROMP is discussed on the basis of their structural differences. A detailed NMR study of the reaction of Zr-1, Zr-2, Hf-1 and in particular of dimethylsilylene-bis(6-[2-(diethylboryl)phenyl]pyrid-2-ylamido)ZrCl2 (Zr-4) with MAO and with MAO/NBE clarifies the reaction cascade from VIP to ROMP and allows for establishing a general concept for a tailored switch from VIP to ROMP within the same polymer chain, allowing for tandem VIP–ROMP copolymerization of E with NBE.

 Please wait while we load your content...
Please wait while we load your content...