Bifunctional polybenzoxazine nanocomposites containing photo-crosslinkable coumarin units and pyrene units capable of dispersing single-walled carbon nanotubes†
Abstract
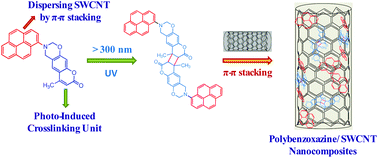
In this study, we have synthesized a new bifunctional benzoxazine monomer (coumarin–Py BZ) possessing both coumarin and pyrene groups through the reaction of 4-methyl-7-hydroxycoumarin (coumarin–OH), paraformaldehyde, and amino-pyrene (Py–NH2) in 1,4-dioxane. Fourier transform infrared (FTIR) and 1H and 13C nuclear magnetic resonance spectroscopy confirmed the structure of this new coumarin–Py BZ monomer. We used differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and FTIR spectroscopy to monitor the curing behavior of coumarin–Py BZ to form poly(coumarin–Py BZ), both before and after photodimerization of the coumarin moiety. DSC profiles revealed that the glass transition temperature of poly(di-coumarin–Py BZ) was higher than that of its corresponding polymer (poly(coumarin–Py BZ)), and consistent with an increase in crosslinking density after UV irradiation. The pyrene moiety of coumarin–Py BZ enhanced the dispersibility of single-walled carbon nanotubes (SWCNTs) in THF, leading to the formation of highly dispersible coumarin–Py BZ/SWCNT nanocomposites stabilized through π–π stacking between the pyrene and SWCNT units, as detected by fluorescence emission spectroscopy. The combination of photo-crosslinkable coumarin groups and SWCNT nanohybrids enhanced the glass transition temperature, thermal stability, and char yield of the polybenzoxazine matrix, based on DSC and TGA analyses.

 Please wait while we load your content...
Please wait while we load your content...