Microporous spiro-centered poly(benzimidazole) networks: preparation, characterization, and gas sorption properties†
Abstract
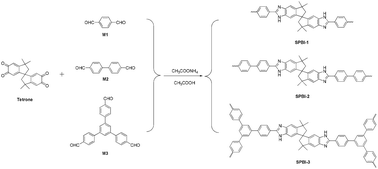
Three microporous spiro-centered poly(benzimidazole) networks were synthesized based on the condensation of di-/trialdehyde with 3,3,3′,3′-tetramethyl-1,1′-spirobisindane-5,5′,6,6′-tetrone in refluxing glacial acetic acid containing ammonium acetate. Fourier transform infrared and solid-state cross-polarization/magic-angle-spinning 13C NMR spectroscopy techniques were utilized to confirm the presence of a benzimidazole ring in the obtained polymers. The morphology can be observed from scanning electron microscopy and transmission electron microscopy images. The materials possess a Brunauer–Emmet–Teller specific surface area ranging from 520 to 600 m2 g−1. The highest hydrogen and carbon dioxide sorption capacity values of the obtained poly(benzimidazole) networks are up to 1.60 wt% (77 K and 1.0 bar) and 13.6 wt% (273 K and 1.0 bar), respectively, which are comparable to those of most of the microporous organic polymers.

 Please wait while we load your content...
Please wait while we load your content...