Improvement of the control over SARA ATRP of 2-(diisopropylamino)ethyl methacrylate by slow and continuous addition of sodium dithionite†
Abstract
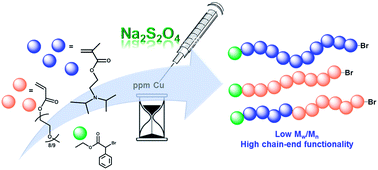
The kinetics and detailed mechanism of SARA ATRP of 2-(diisopropylamino)ethyl methacrylate (DPA) were investigated. Supplemental activator and reducing agent (SARA) atom transfer radical polymerization (ATRP) using sodium dithionite (Na2S2O4) was used to create well controlled polymers of PDPA. The influence of the initiator, solvent, structure and concentration of the catalyst was studied, and the ratios of Na2S2O4 were adjusted to optimize the polymerization. Well controlled polymers required Na2S2O4 to be slowly and continuously fed to the reaction mixture, with 500 parts per million (ppm) of CuBr2 with tris(2-dimethyamino)amine (Me6TREN) as a ligand. The initial content of Na2S2O4 in the reaction mixture, the feeding rate and the Cu catalyst concentration were optimized to provide polymers with narrow molecular weight distribution (Mw/Mn < 1.15) at high monomer conversion (∼90%). Interestingly, the results revealed that when tris(2-pyridylmethyl)-amine (TPMA) was used as a ligand, the amount of copper required to achieve similar control of the polymerization could be decreased 5 times. This system was successfully extended to the polymerization of oligo(ethylene oxide) methyl ether methacrylate (OEOMA). The high conversion and preservation of the chain-end functionality allows the direct synthesis of POEOMA-b-PDPA block copolymers. The low catalyst concentrations and benign nature of Na2S2O4 make this SARA ATRP method attractive for the synthesis of well controlled water soluble polymers for biomedical applications.

 Please wait while we load your content...
Please wait while we load your content...