Mesogen-jacketed liquid crystalline polymers with peripheral oligo(ethylene oxide) chains: phase structure and thermoresponsive behavior†
Abstract
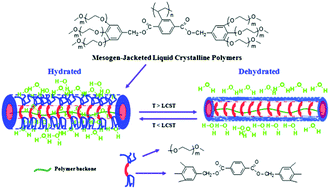
A series of mesogen-jacketed liquid crystalline polymers carrying different repeating unit numbers of oligo(ethylene oxide) (EO) groups (m = 1, 2, 3) and numbers of EO terminal chains (mono, di and tri) have been prepared via free radical polymerization, namely poly{2,5-bis[(4-methoxyoligo(oxyethylene)benzyl)oxycarbonyl]styrene}(P-mono-mEOBCS), poly{2,5-bis[(3,5-dimethoxyoligo(oxyethylene)benzyl)oxycarbonyl]styrene} (P-di-mEOBCS), and poly{2,5-bis[(3,4,5-trimethoxyoligo(oxyethylene)benzyl)oxycarbonyl]styrene} (P-tri-mEOBCS). The chemical structures of the monomers were confirmed by 1H NMR, 13C NMR and mass spectrometry. The characterization of the polymers was performed by 1H NMR, GPC and TGA. The phase structures and transition behaviors were studied using DSC, POM and 1D/2D WAXD. The thermoresponsive behavior was investigated by turbidity measurements using UV-vis spectroscopy. The results showed that P-mono-mEOBCS formed the isotropic phase and was not water-soluble. For P-di-mEOBCS, when m = 1, the polymer exhibited the hexagonal columnar (ΦH) phase and was insoluble. When m = 2, it presented ΦH phases and the lower critical solution temperature (LCST) of the polymer was 23 °C (10 mg mL−1). When m = 3, it showed the nematic columnar (ΦN) phase and the LCST was 40 °C. For P-tri-mEOBCS, all polymers exhibited the ΦH phase and the LCST increased from 22 to 60 °C with increasing m. All of those indicated that the jacking effect and hydrophilicity of polymers increased with increasing numbers of EO peripheral chains. In the meantime, the jacking effect decreased and the hydrophilicity increased with the increase of the EO chain length.

 Please wait while we load your content...
Please wait while we load your content...