Novel hydroxyl-containing reduction-responsive pseudo-poly(aminoacid) via click polymerization as an efficient drug carrier†
Abstract
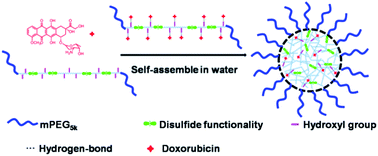
A novel biodegradable pseudo-poly(aminoacid) copolymer (mPEG–HRSCP–mPEG) was produced via a mechanism of step polymerization through Cu(I)-catalyzed azide–alkyne cycloaddition (CuAAC) (click polymerization) using dialkyned-cystine and diazide monomers. The disulfide and hydroxyl groups were repeatedly arranged in the polymer backbone. The introduced hydroxyl groups could not only adjust the hydrophilicity of the amphiphilic polymer, but also improve the DOX loading efficiency and solubility. Compared with the unmodified one, the polymer containing hydroxyl groups has increased hydrophilicity with a larger critical aggregation concentration (CAC) value, lower water contact angle, higher drug loading content and drug loading efficiency. The pH and reduction sensitivities of this drug delivery system (DDS) were investigated using Dynamic Light Scattering (DLS) to monitor the changes of average diameters. DOX was loaded as a model drug and in vitro release experiments demonstrated that DOX release was accelerated by an addition of 10 mM glutathione (GSH) or under acidic conditions rather than under physiological conditions. Our study on in vitro anticancer efficiency showed that DOX-loaded nanoparticles had a higher cytotoxicity towards GSH pretreated HeLa cells. These newly designed copolymer nanoparticles can be used as novel and impactful pH and reduction dual-responsive nanocarriers for intelligent DOX delivery.

 Please wait while we load your content...
Please wait while we load your content...