An enzymatic biomimetic system: enhancement of catalytic efficiency with new polymeric chiral ionic liquids synthesised by controlled radical polymerisation†
Abstract
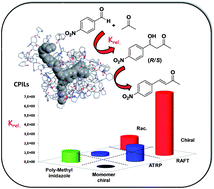
The controlled radical polymerization of monomeric units containing chiral ionic liquids (CILs) allows the synthesis of intrinsically chiral polymers through a bottom-up design. These polymeric chiral ionic liquids (PCILs) show a well-defined three dimensional structure organized as the result of a complex non-covalent network of hydrogen-bonding contacts driven by the C2 hydrogen atoms of the imidazolium aromatic rings and the OH moieties present in the side functionalities of the main polymeric chain. The exchange of the chloride counter ion by the L-prolinate anion leads to new types of polymeric catalysts, which behave as efficient artificial aldolase biomimetic systems, being highly active and selective for the aldol reaction in water. These new polymeric catalysts are significantly more active than the corresponding monomeric counterpart when the reaction is performed either in water or in the presence of water. The increase in catalytic efficiency can be related to their 3D structure, displaying helical chirality in the polymeric chain as a function of their preparation methodology. Under suitable experimental conditions, these polymers are able to catalyse the consecutive aldol-dehydration process, behaving as synthetic mimics of the aldolase-dehydrogenase enzymatic system. Moderate enantioselectivities can be achieved under suitable conditions.

 Please wait while we load your content...
Please wait while we load your content...