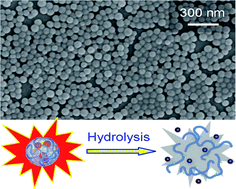
A facile approach for the synthesis of hyperbranched polyacrylates is developed. Tetraphenylethene-containing di- and tetra-acrylates are synthesized by esterification of bis(4-hydroxylphenyl)diphenylethene and tetrakis(4-hydroxylphenyl)ethene with acryloyl chloride and polymerized using azobisisobutyronitrile in refluxing THF, furnishing high molecular weight (Mw up to 87 100) polyacrylates with glycogen-like structures and closed-loops in nearly quantitative yields (isolation yield up to 99%). All the polymers are soluble in common organic solvents and are thermally stable with degradation temperatures up to 357 °C under nitrogen. Whereas the monomers and polymers are nonemissive or faintly emissive in solutions, they become strong emitters in the aggregated state, demonstrating a phenomenon of aggregation-induced or enhanced emission. The emission of the monomer and polymer spots on the TLC plates can be turned “off” and “on” continuously and reversibly by wetting and dewetting processes by vapors of organic solvents. The polymers are photosensitive and UV irradiation of their films through copper masks crosslinks the exposed parts, generating fluorescent negative photoresist patterns. The polymers possess good optical transparency and show high refractive indices (RI = 1.6825–1.5653) in a wide wavelength region (400–1700 nm) with low chromatic dispersion. The emission of the polymer nanoaggregates can be quenched efficiently by picric acid with large quenching constants up to 2.07 × 105 M−1, suggesting that they are sensitive fluorescent sensors for explosive detection. The fluorescent polymers become weakly emissive when hydrolyzed under appropriate conditions, implicative of their utility in controlled drug delivery.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...