Copper(i)–polymers and their photoluminescence thermochromism properties†
Abstract
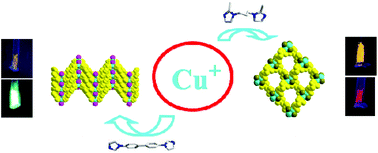
Under hydro(solvo)thermal conditions, four organic bidentate bridging N,N′-donor ligands 1,3-bis(2-methylimidazol-1-yl)propane (L1), 4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl (L2), 1,2-bis(2-methyl-1H-imidazol-1-ylmethyl)benzene (L3) and 5,6,7,8-tetrahydroquinoxaline (L4) were employed to react with CuBr/CuI, generating four 2-D layered copper(I)–polymer coordination polymer materials [Cu2Br2(L1)] 1, [CuI(L2)] 2, [CuI(L3)] 3 and [CuI(L4)0.5] 4. In 1–4, different Cu–X motifs are found: a cubic Cu4Br4 core in 1; a castellated Cu–I single chain in 2; a rhombic Cu2I2 core in 3; and a staircase-like Cu–I double chain in 4. The 2-D layer networks of 1–3 can all be simplified into a simple 44 topology (planar for 1 and 3; wave-like for 2), while the 2-D layer network of 4 has a 63 topology. The photoluminescence behaviors of 1–4 under a UV lamp suggest that 1 and 2 possess fluorescence thermochromism properties. Under the UV lamp, with the decrease in temperature, (i) 1 exhibits a yellow-to-red emission; (ii) 2 exhibits a yellow-to-green emission; (iii) 3 always emits green light; and (iv) 4 never emits light. These are further confirmed by their emission spectra. From 297 K to 77 K, the emission of 1 exhibits a large red shift from 561 nm to 623 nm; the emission of 2 exhibits a large blue shift from 571 nm to 515 nm; only a minor red shift is observed for the emission of 3; and no peaks appear in the emission spectra of 4. The crystal data of 1 and 2 at different temperatures have been collected for revealing the origination of their fluorescence thermochromism properties. Based on the above investigations, the effect of the rigidity/flexibility of the organic ligand on the fluorescence thermochromism properties of copper(I)–polymer coordination polymer materials is discussed. The quantum yields at 297 K and the photoluminescence lifetimes at 297 K and 77 K for 1–3 were also measured for better understanding their photoluminescence properties.


 Please wait while we load your content...
Please wait while we load your content...