Photophysical properties of hexyl diethylaminohydroxybenzoylbenzoate (Uvinul A Plus), a UV-A absorber†
Abstract
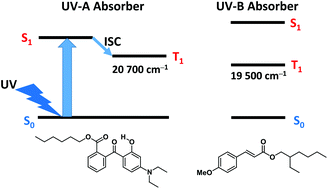
Hexyl diethylaminohydroxybenzoylbenzoate (DHHB, Uvinul A Plus) is a photostable UV-A absorber. The photophysical properties of DHHB have been studied by obtaining the transient absorption, total emission, phosphorescence and electron paramagnetic resonance spectra. DHHB exhibits an intense phosphorescence in a hydrogen-bonding solvent (e.g., ethanol) at 77 K, whereas it is weakly phosphorescent in a non-hydrogen-bonding solvent (e.g., 3-methylpentane). The triplet–triplet absorption and EPR spectra for the lowest excited triplet state of DHHB were observed in ethanol, while they were not observed in 3-methylpentane. These results are explained by the proposal that in the benzophenone derivatives possessing an intramolecular hydrogen bond, intramolecular proton transfer is an efficient mechanism of the very fast radiationless decay from the excited singlet state. The energy level of the lowest excited triplet state of DHHB is higher than those of the most widely used UV-B absorbers, octyl methoxycinnamate (OMC) and octocrylene (OCR). DHHB may act as a triplet energy donor for OMC and OCR in the mixtures of UV-A and UV-B absorbers. The bimolecular rate constant for the quenching of singlet oxygen by DHHB was determined by measuring the near-IR phosphorescence of singlet oxygen. The photophysical properties of diethylaminohydroxybenzoylbenzoic acid (DHBA) have been studied for comparison. It is a closely related building block to assist in interpreting the observed data.


 Please wait while we load your content...
Please wait while we load your content...