A selective emissive chromogenic and fluorogenic seleno-coumarin probe for Cu2+ detection in aprotic media†
Abstract
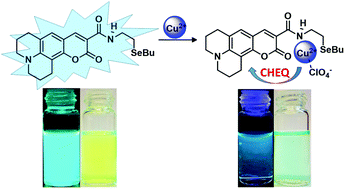
A new selenium containing coumarin (compound 7) was designed and synthesized from the amide linkage between coumarin-519 (6) and 2-(butylselanyl)ethanamine (5). The molecular structure of 7 was accurately characterized, and its photophysical properties in acetonitrile, ethanol and chloroform solutions were studied by absorption, stationary and time-resolved fluorescence spectroscopies. Changes in the solvent polarity affected the Stokes shift, quantum yields and lifetime of the excited states. The spectroscopic behavior of compound 7 was evaluated in the presence of different monovalent, divalent and trivalent metallic cations (Na+, K+, Ca2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+, Pb2+, Hg2+, Hg+, Ag+, Al3+, Fe3+, Ga3+ and Cr3+) in acetonitrile solution. Among the tested cations, 7 exhibited high selective interaction with Cu2+, which was evidenced by the not expected absorption hypsocromic shift (usually coumarin-519 gives red-shifted complexes) and intense chelation-enhanced fluorescence quenching (CHEQ). We performed spectrophotometric and spectrofluorimetric titrations of 7 upon addition of Cu2+. From these data, the minimal detectable and quantifiable amounts were calculated and found to be 0.2 and 0.4 μmol L−1 by absorption and 0.6 and 1.0 μmol L−1 by emission, respectively. The 7-Cu2+ compound presented the 1 : 1 stoichiometry and the stability constant values of absorption and emission were found to be log β = 5.78 and log β = 6.32 respectively. Taking into account the high selectivity of the 7-Cu2+ compound in organic solvent systems, and considering the role of copper in organic transformations, it can be regarded as a promising fluorescent sensor for studies concerning the determination of oxidation-dependent transient entities in organic reactions like those involving cuprates. Additionally, it can be used for the detection and quantification of this metal cation in vitro in aprotic biological systems.


 Please wait while we load your content...
Please wait while we load your content...