Anthracene photodimerization in a bis-chromophoric hydrogen-bonding receptor†
Abstract
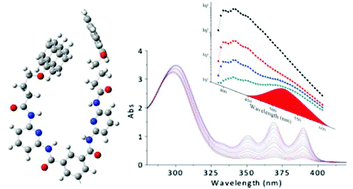
The photochemistry and photophysics of a bis-anthracenyl hydrogen-bonding receptor (7-(3,5-bis((6-(4-(anthracen-9-yloxy) butanamido)pyridin-2-yl)carbamoyl)phenylhept-6-ynoate, 2) were investigated in a fluid solution. At room temperature, fluorescence and intersystem crossing compete with intramolecular dimerization to form head-to-head and head-to-tail photoproducts, whereas fluorescence is the principal deactivation pathway at low temperature. Time-resolved fluorescence spectroscopy evidences two excimer-like emitting states whose sensitivity to quenching by oxygen is considerably lower than that of intramolecular photodimerization (KSV = ca. 450 vs. <100 L mol−1, respectively). Based on the temperature-dependence and quenching efficiency of photodimerization, a mechanism is proposed in which the ground-state conformation of the tether controls the formation of the HH vs. HT photodimers via a non-emissive intermediate.
- This article is part of the themed collection: Dedicated to the memory of Prof. Nicholas J. Turro

 Please wait while we load your content...
Please wait while we load your content...