Tin(ii)-catalyzed dehydrative cross-coupling of 2H-chromene hemiacetals with ketones†
Abstract
A dehydrative cross-coupling of 2H-chromene hemiacetals with ketones is described. Without the derivatization of 2H-chromene hemiacetals to 2H-chromene acetals, the direct C–OH/C–H coupling reaction has been accomplished with water as the only by-product. With the use of Sn(OTf)2 as the promoter, the reaction goes smoothly under mild conditions.
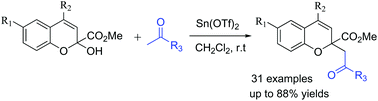
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...