Regioselective deoxygenative chalcogenation of 7-azindole N-oxides promoted by I2/PEG-200†
Abstract
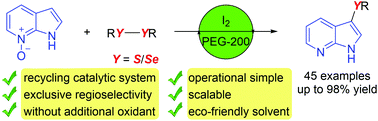
We developed a general and sustainable approach for the regioselective deoxygenative chalcogenation of 7-azindole N-oxides; the combination of an internal oxidant and a green solvent has been used successfully for the synthesis of mono- and dichalcogenyl 7-azaindoles which are of pharmaceutical interest. The regioselectivity is tunable by the variation of the reaction conditions. I2/PEG was established as an efficient and reusable catalytic system for C–H chalcogenation. This developed methodology has great potential for practical utility, with a broad substrate scope, green reaction conditions, and operational simplicity.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...