Modifications of amino acids using arenediazonium salts
Abstract
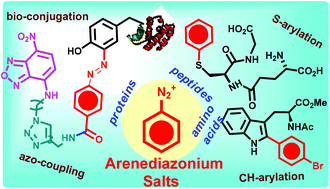
Aryl transfer reactions from arenediazonium salts have started to make their impact in chemical biology with initial forays in the arena of arylative modifications and bio-conjugations of amino acids, peptides and proteins. The unique multimodal reactivity of arenediazonium salts, ranging from thermal or photochemical radical chain reactions, Pd-catalyzed coupling to arylazo-coupling reactions, all under distinct but mild conditions, provides multiple options for side chain modifications of amino acids and peptides and in addition, site-selective protein conjugation and labelling, protein immobilization, azo-bridged macrocyclization, etc. under bio-ambient conditions. The purpose of this review is to highlight these recent advances and to stimulate interest towards broader applications of arenediazonium salts as aryl transfer agents in bioconjugation reactions.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...