Cooperativity and serial ligand catalysis in an allylic amination reaction by Pd(ii)-bis-sulfoxide and Brønsted acids†
Abstract
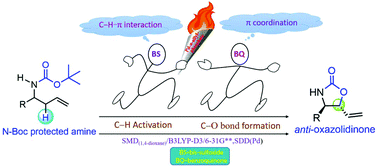
In recent years, transition metal catalysts have been increasingly employed in conjunction with Brønsted acids under one-pot reaction conditions, opening up newer avenues for dual catalytic protocols. Under such dual catalytic conditions, the general premise of holding the native ligands on the catalyst in the same manner throughout the catalytic cycle becomes immediately questionable. We have invoked the likelihood of Serial Ligand Catalysis in an important intramolecular allylic amination of N-Boc (N-tert-butoxycarbonyl) protected homoallylic amine leading to an anti-oxazolidinone product. The reported reaction conditions employed (bis-sulfoxide)Pd(OAc)2 and dibutyl phosphoric acid (DBPOH) as the catalysts and benzoquinone (BQ) as the oxidant. We used density functional theory computations at the B3LYP-D3 level of theory to examine a comprehensive set of ligand combinations around the Pd center so as to identify the energetically most preferred pathway. The key catalytic events consist of (i) a C–H activation at the allylic position in the catalyst–substrate complex [Pd(L)(L′)2(substrate)], leading to a (L)(L′)Pd–π-allyl intermediate, and (ii) an intramolecular C–O bond formation between the carbonyl oxygen of the N-Boc amine and the allyl carbon. Interesting cooperativity between the catalysts in both these steps has been found, wherein the Pd(DBPO−)2(BS) species is involved in the C–H activation transition state and Pd(DBPO−)(BQ) in the C–O bond formation step. The energetic advantage in swapping the bis-sulfoxide ligand on Pd with a benzoquinone upon moving from the first step to the second step confirms the significance of serial ligand catalysis in dual catalytic reactions.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...