Synthesis and biological evaluation of fluorinated analogues of ripostatin A†
Abstract
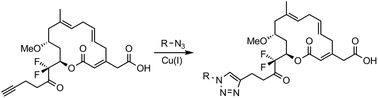
Several monocyclic derivatives of 14,14′-difluororipostatin A were prepared using a catalytic Mukaiyama aldol reaction, a ring-closing metathesis reaction and a late stage click reaction as key steps. The biological activity of the produced compounds was assessed in vivo using a panel of pathogenic microorganisms. Moderate antibiotic activity was observed for 11-OMe-ripostatin A and 11-OMe-14,14′-difluororipostatin A.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...