1,8-Diamidocarbazoles: an easily tuneable family of fluorescent anion sensors and transporters†
Abstract
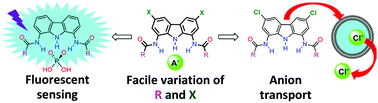
The synthesis, structure and anion recognition properties of an extensive, rationally designed series of bisamide derivatives of 1,8-diaminocarbazole and 1,8-diamino-3,6-dichlorocarbazole are described. Despite simple structures and the presence of only three hydrogen bond donors, such compounds are remarkably strong and selective receptors for oxyanions in DMSO + 0.5%H2O. Owing to their carbazole fluorophore, they are also sensitive turn-on fluorescent sensors for H2PO4− and AcO−, with a more than 15-fold increase in fluorescence intensity upon binding. Despite relatively weak chloride affinity, some of the diamidocarbazoles have also been shown, for the first time, to be very active chloride transporters through lipid bilayers. The binding, sensing and transport properties of these receptors can be easily modulated by the usually overlooked variations in the length and degree of branching of their alkyl side arms. Overall, this study demonstrates that the 1,8-diamidocarbazole binding unit is a very promising and synthetically versatile platform for the development of fluorescent sensors and transporters for anions.


 Please wait while we load your content...
Please wait while we load your content...