A transition state “trapped”? QM-cluster models of engineered threonyl-tRNA synthetase†
Abstract
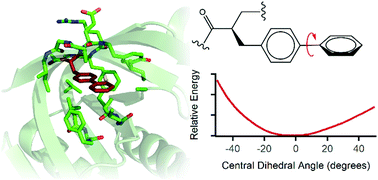
In a recent study [Science, 2015, 347, 6224], protein engineering was used to design a core within the enzyme threonyl-tRNA synthetase (ThrRS) capable of stabilizing the coplanar transition state conformation of an inserted noncanonical p-biphenylalanine (BiPhe) residue. Using the X-ray crystal structures of the preliminary (Protein Data Bank entries 4S02, 4S0J, 4S0L, 4S0I, and 4S0K) and final (PDB entry 4S03) ThrRS proteins, fully quantum mechanical (QM) cluster models were constructed and analyzed. Density functional theory and molecular dynamics computations were performed to investigate the energetic profiles of BiPhe dihedral rotation within the ThrRS models. For the 4S03 model, results indicate that steric and hydrophobic forces of the residues surrounding BiPhe eliminate the coplanar transition state entirely. Molecular dynamics simulations were carried out that confirmed the extent of BiPhe rotational flexibility, and provided additional information on barrier heights of full BiPhe rotation. Transition states of near-coplanar biphenyl rings of BiPhe were found for the 4S0I and 4S0K models, but are not likely persistent on any observable timescale. The dihedral angle of the biphenyl moiety is thermally allowed to fluctuate within the ThrRS protein core models by a range of 17°–26°. BiPhe–residue interaction counts (RICs) were used to compare the interaction differences among the different ThrRS cores. The RICs demonstrate how BiPhe is compacted within the 4S03 core, resulting in the experimentally observed “trapped” coplanar transition state analogue. This work presents a unique application of QM-cluster models towards studying the inner workings of proteins, and suggests avenues that computational chemistry can be used to further guide bioengineering.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...